Current Projects
Visual System Response to Electrical Simulation
The long-term goal of this project is to investigate safe and effective retinal stimulation paradigms. We conduct experiments in animal models, including models of inherited outer retinal degeneration, and in humans with Argus II retinal implants. In animals, we use single-cell electrophysiology and calcium imaging to assess the response at cellular resolution. We also use optical coherence tomography to measure the safety of high-intensity, continuous stimulation. To assess visual receptive fields, we measure visual cortex responses in small animals. Human testing focuses on the shape and other perceptual characteristics of single and multi-channel stimulation.
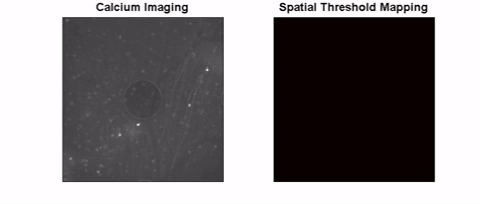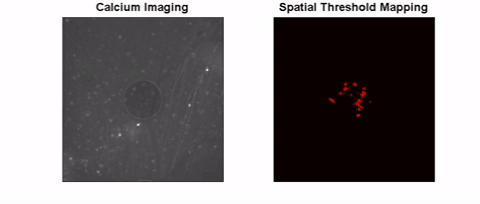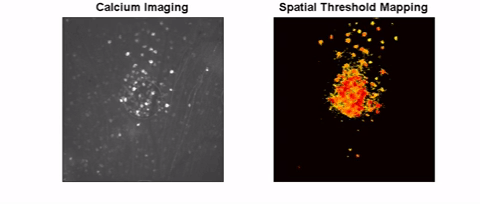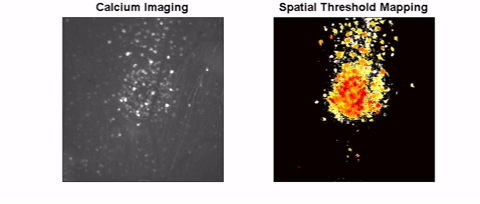
High-Resolution Retinal Prosthesis
We are creating a new approach for retinal prostheses based on carbon fiber electrodes inserted into the retina. This will place the stimulating electrodes near the target cells while avoiding stimulation of retinal ganglion cell axons. Carbon fibers maintain strength even at small diameter (>7 microns). Given the small size of carbon fibers, it is possible to make a stimulating array with a spacing of 40 microns in between each fiber, which can theoretically provide artificial vision better than the threshold for legal blindness (20/200). Small and consistent electrode active sites are also an important factor for producing high-resolution vision. We are investigating methods for shaping CFs tip to optimal sizes for stimulation that will small enough to target only nearby cells but large enough to allow sufficient stimulus current within safe stimulation guidelines. Fabrication methods under study include laser sharpening and chemical etching.
Adaptive Vision Assistant
We are building a multimodal user interface embedded in a glasses frame to serve as an Adaptive Vision Assistant (AVA). The first task we are undertaking is to design AVA as a mobility aid, but other tasks are possible as well. AVA.glass is a headworn user interface. A camera (embedded in AVA.glass) will obtain environmental information for apps that rely on real-time video. AVA.glass is controlled by AVA.app, a smartphone app that serve as an intelligent assistant for the user. AVA.app integrate, by integrating information from multiple sources and automatically launching single task apps when needed. This system will be hands free and will relieve the user from opening and closing multiple apps while also navigating, thus addressing two deficiencies with current assistive technology. To increase user confidence, our system will provide multi-modal input, with information provided via vibrotactile and auditory channels. Our prior work supports the immediate, intuitive nature of vibrotactile cues while verbal descriptions can provide more detailed information. The smartphone will provide necessary resources (computational, sensing, connectivity). Our approach will move only necessary functions of the phone to the glasses, specifically, the camera, sound display, and tactile display, which will constrain cost, improve aesthetics, and delay obsolescence.

Current works within the software components of AVA include a built-in assistant, launching third-party apps such as Google Maps, Be My Eyes, and Seeing AI. Also in the works is obstacle detection and avoidance using the Depth Anything and Segformer model. The app serves as the central control hub of the system, enabling users to interact with their smartphones and navigate their surroundings more independently and efficiently. All features are optimized for real-time, on-device execution and provide feedback through audio cues and directional vibrations via our custom AVA smartglass. By combining intuitive interaction with intelligent perception, AVA aims to enhance both smartphone usability and the safety and efficiency required for mobility.


Retinal Electrophysiology
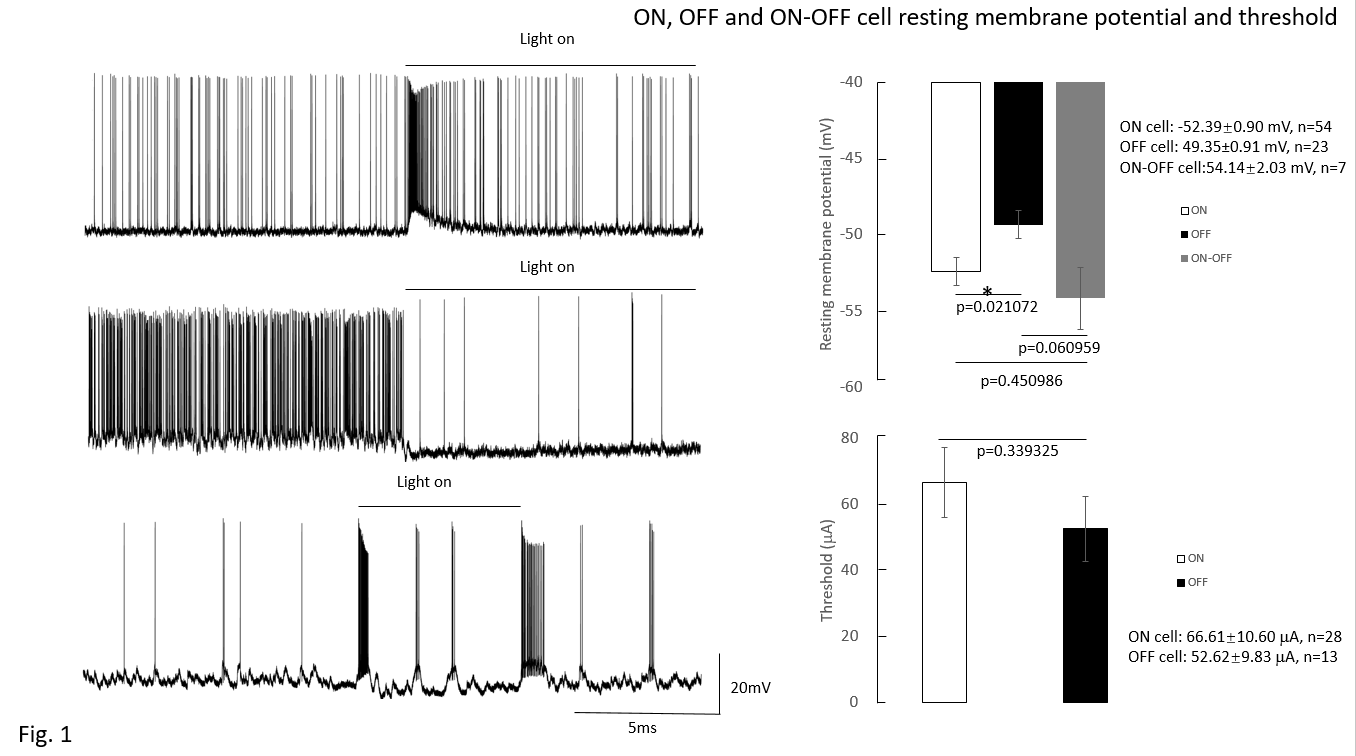
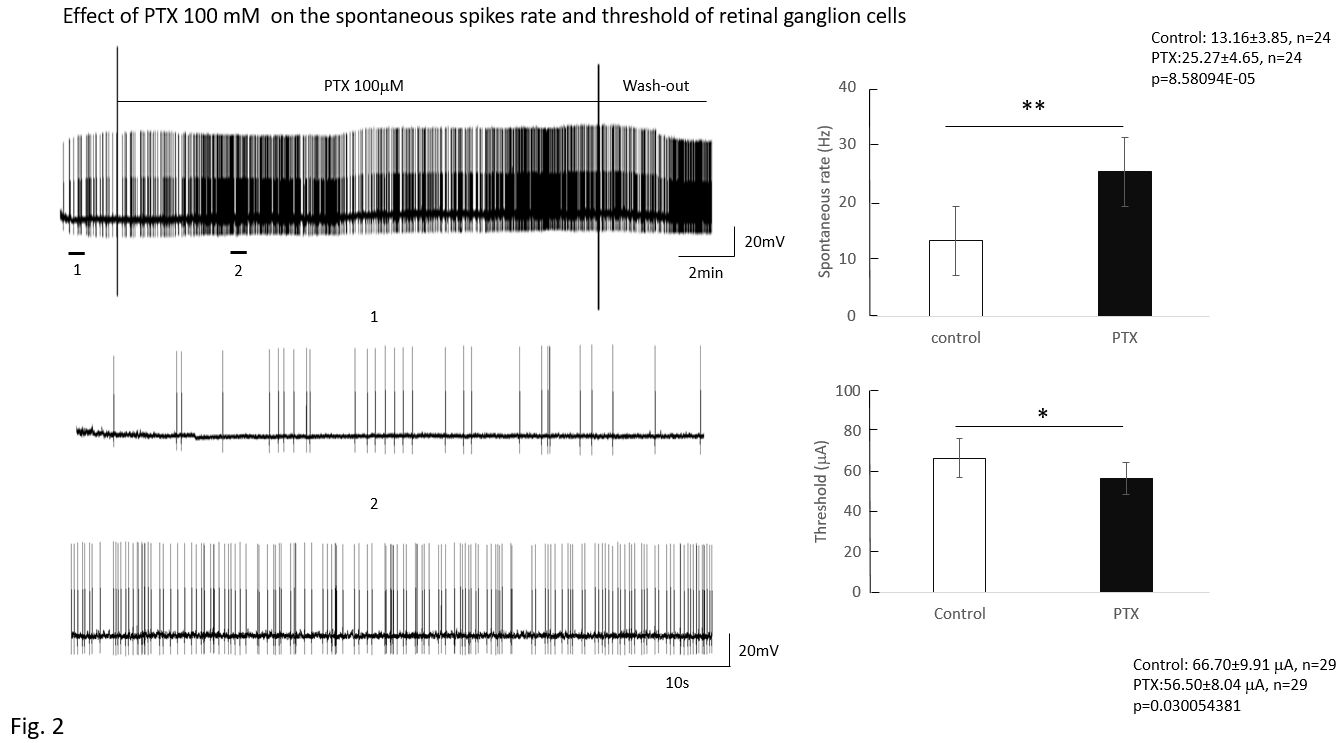
This study focuses on the effect of GABA on the mice retinal ganglion cells (RGCs) to identify the regulatory role of amacrine cells in RGCs. RGCs were classified into ON, OFF, and ON-OFF types using 10-second light exposure. Electrical stimulation pulses were applied via an epiretinal electrode (platinum, 75 micron diameter). Pulses were 0.5 ms, cathodic first, biphasic, with a train of 20 pulses delivered at 2 pulses/second. To obtain response data, pulse amplitude was increased in steps. Then the number of pulses that evoked spikes was counted and threshold was set at the 50% response rate. No difference was noted in threshold based on cell type. After administration of picrotoxin (100 mM), threshold decreased (control: 73.6±15.02mA; 100 mM picrotoxin: 62.36±13.95 mA, n=25, p=0.02782). RGCs spontaneous spikes rate increased after picrotoxin (control: 13.16±3.85 spikes/s; 100 mM picrotoxin: 25.27±4.65 spikes/s, n=24, p=8.581×10-5). These implied that blocking amacrine cells’ GABAa receptor increased the excitability of RGCs as reflected by decreases in action potential threshold.